Search the Newsroom
Filter By
News Type
Related specialties and programs
Showing 1 - 5 of 5 results

FDA Approves First Drug for Friedreich’s Ataxia
The drug, omaveloxolone, is a once-a-day pill that will help improve neurological function and slow progression of the neurodegenerative disease.
Trial Involving CHOP Researchers Shows Drug Helps Pediatric Patients with Hypertrophic Cardiomyopathy
CHOP researchers participated in clinical trial that showed valsartan has a measurable benefit for patients with early-stage hypertrophic cardiomyopathy.
Friedreich’s Ataxia Center of Excellence at CHOP Awarded $1.275 Million to Advance Medical Research
The Friedreich’s Ataxia Center of Excellence at CHOP was awarded $1.275 million by the Friedreich’s Ataxia Research Alliance, Hamilton and Finneran families and the CureFA Foundation
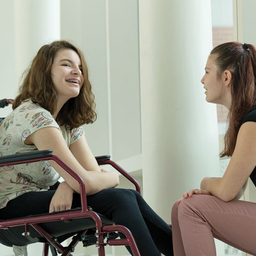
In Pursuit of Cure for a Rare Degenerative Disease
A rare degenerative disease strikes during childhood. A multimillion-dollar gift is fueling CHOP scientists’ search for a cure.

$3.25 Million Gift Creates Friedreich’s Ataxia Center of Excellence
Donations from Friedreich’s Ataxia Research Alliance and the Hamilton and Finneran Families helps launch Penn Medicine/CHOP Center to improve research and care for Friedreich's Ataxia, a rare, degenerative disease.